THE SPIKE PROTEIN, COVID-19, PASC, DEMYELINATION AND ANTIPHOSPHOLIPID SYNDROME
August 3, 2022

While looking for a more satisfactory and complete explanation for the observed autoimmunity, sudden cardiac deaths, high altitude incidents and demyelination occurring I decided to “look one move deeper” in my calculations of the mechanisms involved in Spike Protein pathology. My work has revealed that the “man behind the curtain” is the Spike Protein’s ability to literally remove lipids (by binding) from membranes and induce Lipid Peroxidation. These mechanisms can explain the etiology of the aforementioned autoimmune diseases, thromboembolic events and conditions.
LIPID BINDING AND SPIKE PROTEIN IMMUNE COMPLEXES
A very curious quality of the Spike Protein is its ability to REMOVE lipids from membranes.
Our studies suggest that the SARS-CoV-2 spike protein binds to model membranes and removes lipids, and potentially the process is facilitated by the presence of cholesterol.
SARS-CoV-2 spike protein removes lipids from model membranes and interferes with the capacity of high density lipoprotein to exchange lipids
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8195693/
However, nobody appears to be concerned with the potential autoimmune implications of this phenomenon. If you have Spike Proteins circulating or being presented to APCs which are coated with the body’s own lipids, there is a very high likelihood of autoantibodies being induced.
THE SPIKE PROTEIN ALONE INDUCES LIPID PEROXIDATION

In addition to the Spike Protein’s ability to bind and remove lipids from membranes, it also has the ability to induce Lipid Peroxidation on its own, independent of viral infection. Lipid Peroxidation is the chain of reactions of oxidative degradation of lipids. It is the process in which free radicals "steal" electrons from the lipids in cell membranes, resulting in cell damage. This process proceeds by a free radical chain reaction mechanism.
Here we show that S protein stimulation for 24 hours, independently of hypoxia, induced lipid peroxidation associated to oxidative stress in pericytes. Additionally, our data indicate that hypoxia exacerbated the S protein-mediated the generation of ROS and RNS in brain pericytes. The results suggest that S protein is sufficient to trigger oxidative and nitrosative stress in pericytes, which is exacerbated under hypoxic conditions.
SARS-CoV-2 spike protein induces brain pericyte immunoreactivity in absence of productive viral infection
https://www.biorxiv.org/content/10.1101/2021.04.30.442194v1.full
The phenomenon is not limited to pericytes.
In this study, we attempted to explore the direct interplays among the Spike protein, lipid metabolism, autophagy, and ferroptosis in host cells. We found that the Spike protein impairs lipid metabolic and autophagic pathways in host cells, leading to increased susceptibility to lipotoxicity most likely via switching on nuclear factor erythroid 2-related factor 2 (Nrf2)-mediated ferroptosis. Ferroptosis is an iron-dependent lipid peroxidation-driven cell death.
The Spike Protein of SARS-CoV-2 Impairs Lipid Metabolism and Increases Susceptibility to Lipotoxicity: Implication for a Role of Nrf2
https://www.mdpi.com/2073-4409/11/12/1916/htm
INDUCTION OF ANTIPHOSPHOLIPID SYNDROME
Through the mechanisms discussed above, I believe that Antiphospholipid Syndrome is being induced by the Spike Protein.
The mechanism leading to the formation of antiphospholipid antibodies (aPL) is still unknown. Because an in vitro study suggested that aPL may derive from pro-oxidant conditions, we sought a relationship between aPL and isoprostanes, indices of lipid peroxidation in vivo. This study, showing the existence of a close association between aPL and increased in vivo lipid peroxidation, supports the hypothesis that these antibodies may result from pro-oxidative conditions and suggests that inflammation may play an important role.
Enhanced Lipid Peroxidation in Patients Positive for Antiphospholipid Antibodies
https://ashpublications.org/blood/article/90/10/3931/139483/Enhanced-Lipid-Peroxidation-in-Patients-Positive
INDUCTION OF DEMYELINATION
Once the induction of Antiphospholipid Antibodies has occurred, these antibodies then attack myelin structures in the body, causing massive Demyelination.
Neurologic manifestations are common in patients with antiphospholipid antibodies and include stroke, seizures, dementia, cognitive dysfunction, chorea, migraine, psychosis, and demyelinating disease.
Central Nervous System Manifestations of Antiphospholipid Syndrome
https://pubmed.ncbi.nlm.nih.gov/29061241/
And, what have we observed throughout the course of COVID and the presence of Spike Proteins?
Connective tissue diseases can be characterised by central nervous system (CNS) involvement, in some patients manifested by demyelination areas in the white matter of the brain and spinal cord, which are difficult to differentiate from multiple sclerosis (MS) and other demyelinating processes, such as transverse myelitis and optic neuritis. Demyelinating process may be the feature of nervous impairment in systemic lupus erythematosus, Behcet's disease (BD), Sjoegren's syndrome (SS), systemic sclerosis (SSc) or very rarely other systemic autoimmune diseases.
Non-MS autoimmune demyelination
https://www.sciencedirect.com/science/article/abs/pii/S0303846708002126?via%3Dihub
New or Worsening Neurologic Symptoms Post-COVID in Patients With Multiple Sclerosis
https://www.neurologyadvisor.com/topics/multiple-sclerosis/new-worsening-neurologic-symptoms-post-covid-patients-multiple-sclerosis/
Acute Transverse Myelitis (ATM): An Unexpectedly Frequent Complication of COVID-19
https://www.houstonmethodist.org/leading-medicine-blog/articles/2021/jun/acute-transverse-myelitis-atm-unexpectedly-frequent-in-covid-19-patients-study-finds/
COVID-19-associated optic neuritis – A case series and review of literature
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8917537/
Systemic lupus erythematosus manifestation following COVID-19: a case report
https://jmedicalcasereports.biomedcentral.com/articles/10.1186/s13256-020-02582-8
Behçet’s-like adverse event or inaugural Behçet’s disease after SARS-CoV-2 mRNA-1273 vaccination?
https://academic.oup.com/rheumatology/article-abstract/61/5/e112/6383152
Increase in the number of Sjögren's syndrome cases in Brazil in the COVID-19 Era
https://onlinelibrary.wiley.com/doi/10.1111/odi.13925
Case Report: Systemic Sclerosis After Covid-19 Infection
https://www.frontiersin.org/articles/10.3389/fimmu.2021.686699/full
SUDDEN ATHLETE DEATHS, HIGH ALTITUDE INCIDENTS AND SENESCENCE
The observed sudden deaths in athletes, the incidents of those with long term exposure to high altitudes, such as pilots, and the ubiquitous Senescent cells can ALL be explained by the induction of Antiphospholipid Syndrome.
Impaired aerobic exercise capacity and cardiac autonomic control in primary antiphospholipid syndrome
https://www.researchgate.net/publication/253335894_Impaired_aerobic_exercise_capacity_and_cardiac_autonomic_control_in_primary_antiphospholipid_syndrome
Fifteen sudden death male victims (average age 43.93 + 7.81) years and twenty clinically healthy subjects were investigated for antiphospholipid antibodies (aPLs) - anticardiolipin antibodies (aCL), class IgG and IgM; anti-β2-glycoprotein 1 antibodies (anti-B2-GPI) - class IgG and IgM; total cholesterol and triglycerides. The results obtained lead to an assumption that aPLs play a role in the pathogenesis of acute coronary incident, provoking a sudden cardiac death in young subjects without atherosclerotic vascular changes.
Antiphospholipid antibodies in cases of sudden cardiac death
https://www.researchgate.net/publication/287747204_Antiphospholipid_antibodies_in_cases_of_sudden_cardiac_death
Antiphospholipid syndrome (APS) is an autoimmune clotting disorder that may present catastrophically with multiple thromboses over a short period of time. In this article, we examine the case of a woman with undiagnosed APS whose first symptoms presented during a long-haul flight. A review of the literature on thrombosis at high altitudes and during long duration travel helps us understand potential treatment and prevention of the same in APS patients.
Antiphospholipid Syndrome: The Risk of Travel at High Altitudes
https://www.the-rheumatologist.org/article/antiphospholipid-syndrome-the-risk-of-travel-at-high-altitudes/?singlepage=1&theme=print-friendly
Lipids are key macromolecules that perform a multitude of biological functions ranging from maintaining structural integrity of membranes, energy storage, to signaling molecules. Unsurprisingly, variations in lipid composition and its levels can influence the functional and physiological state of the cell and its milieu. Cellular senescence is a permanent state of cell cycle arrest and is a hallmark of the aging process, as well as several age-related pathologies. Senescent cells are often characterized by alterations in morphology, metabolism, chromatin remodeling and exhibit a complex pro-inflammatory secretome (SASP). Recent studies have shown that the regulation of specific lipid species play a critical role in senescence. Indeed, some lipid species even contribute to the low-grade inflammation associated with SASP. Many protein regulators of senescence have been well characterized and are associated with lipid metabolism.
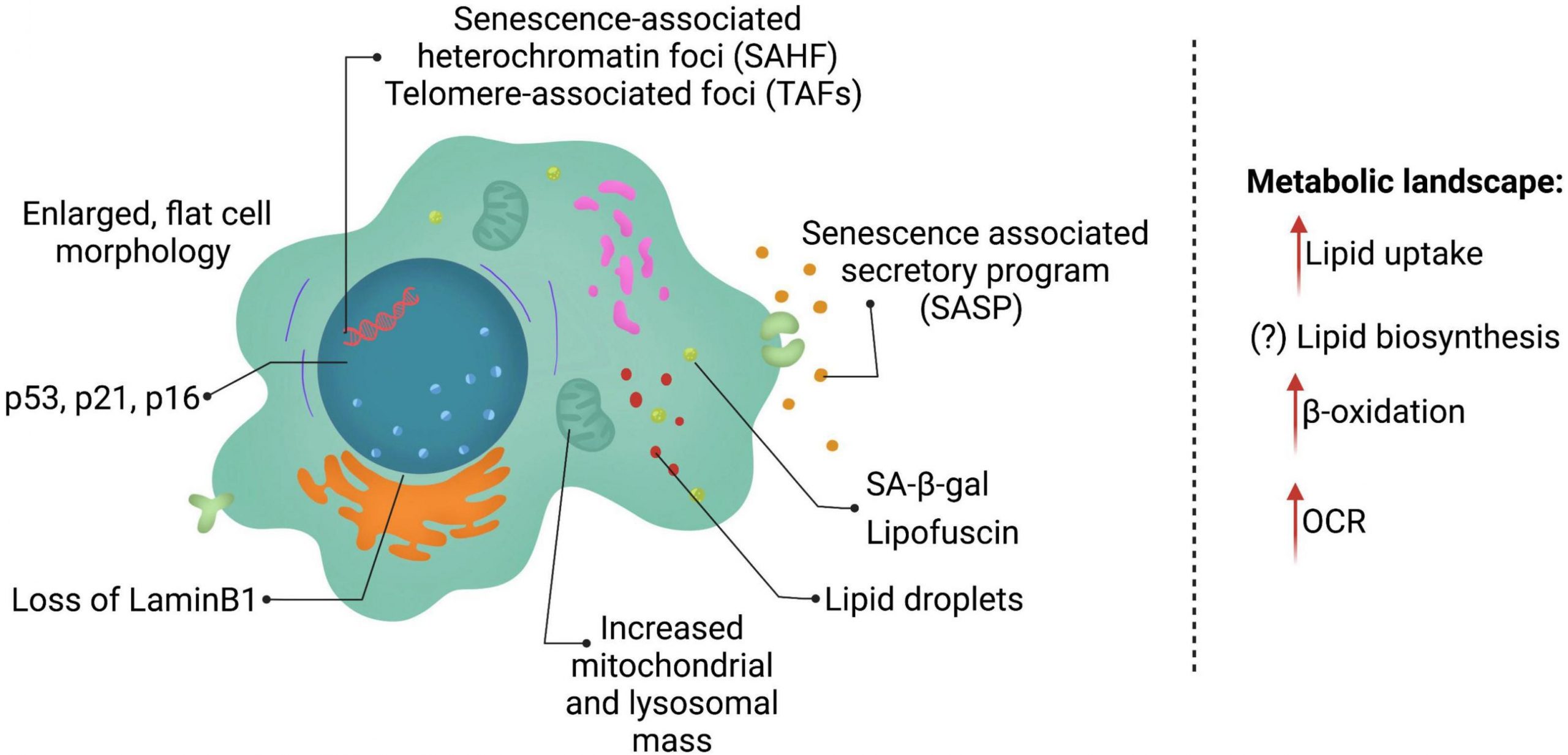
Lipids as Regulators of Cellular Senescence
https://www.frontiersin.org/articles/10.3389/fphys.2022.796850/full
I believe therapeutics directed against Antiphospholipid Antibodies should be trialed for COVID and PASC.
The most important question to ask, if the above is confirmed, is whether repeated exposures to the Spike Protein of SARS-CoV-2 will inexorably result in the victim’s death from Asherson’s Syndrome. This is my greatest concern, and most ominous fear.