June 29, 2021
Concerning iron dysmetabolism in COVID-19, Ehsani has highlighted a remarkable similarity between the distant amino acid sequence of SARS-CoV-2 spike glycoprotein cytoplasmic tail and the hepcidin protein.
Coronaviruses recognize host receptors using their spike proteins, facilitating conformation transition, so to bind cell membrane and enter host cytoplasm; by using host furins and proteases, coronaviruses may cleave their spike polypeptides, thus favoring the cell entry. The found hepcidin mimicry by the virus would take place through this complex mechanism.

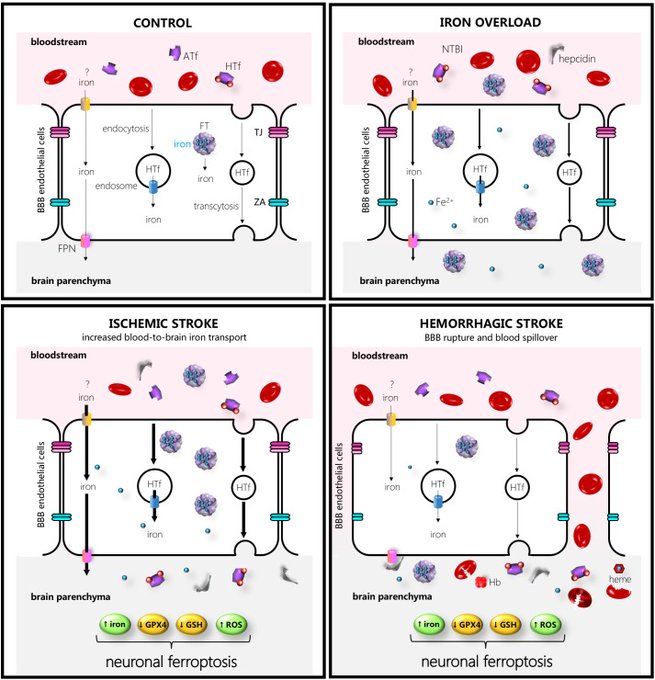
Perhaps the reason we are seeing strokes, blindness and other neurodegenerative activity post spike protein therapies is that that ferroportin is expressed in the endothelial cells of the blood–brain barrier, in neurons, oligodendrocytes, astrocytes, and the choroid plexus and ependymal cells. The cleaved spike protein mimics hepcidin, crossing the blood brain barrier and interacting with ferroportin expressed in the endothelial cells and neurons of the brain.
Additionally, there is evidence that PrPc interacts with hepcidin causing ROS and potentially transforming PrPc to PrPsc.
Referenced/Related Papers
Prions and prion diseases: Insights from the eye
https://sciencedirect.com/science/article/abs/pii/S0014483520304589?via%3Dihub
COVID-19: Hemoglobin, Iron, and Hypoxia beyond Inflammation. A Narrative Review
https://mdpi.com/2039-7283/10/2/1271
Distant sequence similarity between hepcidin and the novel coronavirus spike glycoprotein: a potential hint at the possibility of local iron dysregulation in COVID-19
https://arxiv.org/ftp/arxiv/papers/2003/2003.12191.pdf
Deciphering the Iron Side of Stroke: Neurodegeneration at the Crossroads Between Iron Dyshomeostasis, Excitotoxicity, and Ferroptosis
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6389709/